Attempts to harness the immune system to fight cancer date back to the late 1800s. It has been less than 40 years since the first of these immuno-oncology therapies demonstrated success and obtained approval. Since then, nearly 40 cancer immunotherapies have received FDA approval. These approved treatments fall into five categories of immunotherapy: cell-based, immunomodulators (including checkpoint inhibitors and cytokines), cancer vaccines, antibody-based, and oncolytic viruses. Notable milestones in the immuno-oncology field include the first cancer vaccine, Dendreon Pharmaceuticals’ Sipuleucel T for prostate cancer, approved in 2010, the first checkpoint inhibitor, BMS’ Ipilimumab for metastatic melanoma, approved in 2011, the first oncolytic virus therapy, Amgen’s T-Vec for melanoma, approved in 2015, and the first CAR-T cell therapy, Novartis’ Tisagenlecleucel for acute lymphoblastic leukemia, approved in 2017.
CipherBio PRO Immuno-Oncology Insights
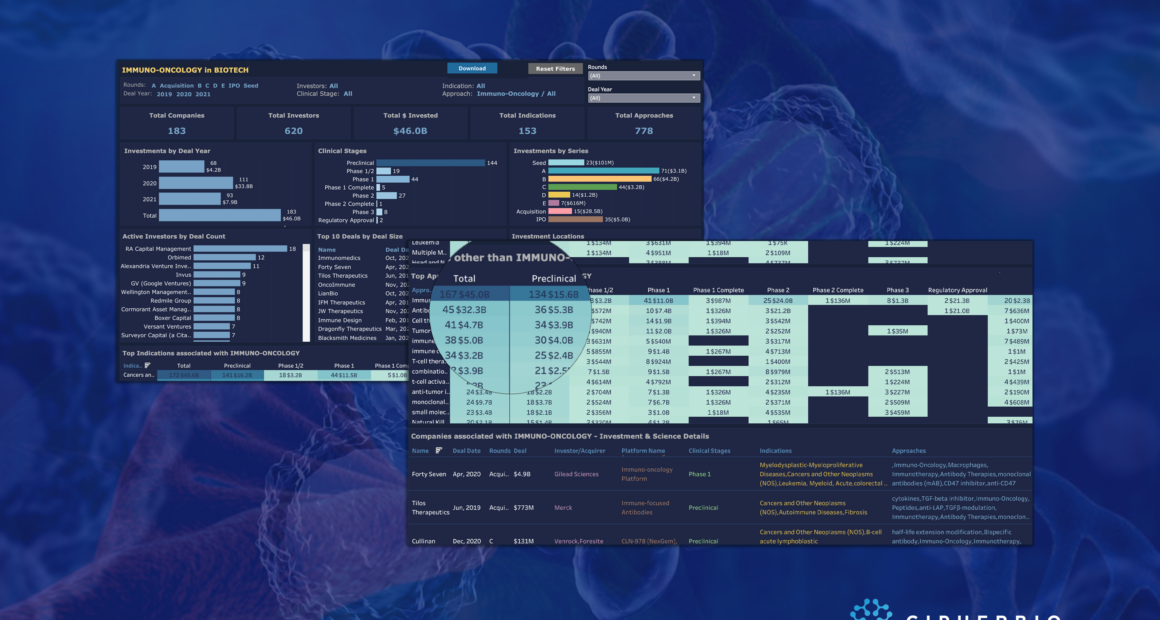
CipherBio PRO Immuno-Oncology Insights explores the immuno-oncology companies that received investments from 2019-2021 and their clinical phases. 183 companies with 620 investors, 153 indications, and over 778 scientific approaches received a total of $46 billion.
Almost half of the $46 billion came from one deal, Gilead’s $21B acquisition of New Jersey-based Immunomedics, a leading antibody-drug conjugate cancer immunotherapy company. Most importantly in this transaction, Gilead received Trodelvy, approved immunotherapy for metastatic triple-negative breast cancer (mTNBC) which accounts for 10-15% of all breast cancer cases.

Antibody Therapies
In addition to Immunomedics, 45 other companies are using antibody-based immunotherapies. They received $32.3B, making antibody therapies the top approach and most common immunotherapy method. Furthest along in clinical trials are Immunmedics (approved Trodelvy) and 2 companies that have product candidates in phase 2. Apexigen Inc. developed Sotigalimab, a CD40 agonist monoclonal antibody currently in phase 2 for soft tissue sarcoma that recently was granted Orphan Drug Designation from the FDA. Apexigen raised a $65 Million Series C extension round in March 2020 led by Decheng Capital and Ocean Pine Capital. NextCure has developed NC318, a monoclonal antibody targeting Siglec-15 (S15) for treatment of S15 expressing cancers including non-small cell lung cancer, head and neck squamous cell carcinoma, ovarian and triple-negative breast cancers. NextCure went public in an $86.2 Million IPO in May 2019 valued at $329 Million with a current valuation of $198.8 Million.

The majority of the antibody therapy companies, 35 of the 45, have products in preclinical trials. Despite not yet having clinical product candidates, Pyxis Oncology had the 2nd biggest investment round of all antibody therapy companies and the biggest of the preclinical antibody therapy companies. In March 2021, Boston-based Pyxis raised a $152 Million Series B led by Arix Bioscience and RTW Investments to support their preclinical work with antibody-drug conjugate and monoclonal antibody immunotherapies for cancer treatment.
Cell Therapies
Cell-based therapies are the second top immuno-oncology method with 41 companies raising $4.7 Billion. Recent advancements and promising results in cancer cell therapy are predominantly due to CAR-T and other t-cell therapies. 29 of the 41 cell therapy companies are using t-cells and 15 of those are using CAR-T cells. However, immuno-oncology cell therapy company Dragonfly Therapeutics is reeling in big funds and does not use the popular t-cells or CAR-T cells. Waltham, MA-based Dragonfly Therapeutics’ platform uses natural killer (NK) cells to treat solid and heme tumors, with their lead NK product candidate in phase 1 trials for HER2 expressing solid tumors and an additional cytokine therapy in phase 1/2 trials. Although the amount of Dragonfly’s most recent investment round in March 2020 was undisclosed, the round brought the company’s total equity to $300 Million, and since then the company has gained massive funds from a $475 Million partnership with BMS and an expansion of a pre-established partnership worth $700 Million with Merck.
Interestingly, Merck partnered with another NK cell company, Artiva Biotherapeutics, to help advance Artiva’s pipeline of CAR-NK immunotherapies. Since its founding at the end of 2018, Artiva raised a $78 Million Series A in June 2020 and went on to raise $120 Million Series B in February 2021 led by Venrock Healthcare Capital Partners. The company’s lead product candidate has already begun clinical testing in a phase 1/2 trial for patients with relapsed B-cell non-Hodgkin lymphoma (NHL).
IPOs
 35 companies went public from 2019-2021 through deals totaling $5 Billion. 30+ of the companies have products in clinical trials including two in phase 3 trials. CStone Pharmaceuticals, which went public in February 2019 through a $285 Million IPO, is the only company with FDA-approved products. 24 of the companies that went public are still preclinical.
35 companies went public from 2019-2021 through deals totaling $5 Billion. 30+ of the companies have products in clinical trials including two in phase 3 trials. CStone Pharmaceuticals, which went public in February 2019 through a $285 Million IPO, is the only company with FDA-approved products. 24 of the companies that went public are still preclinical.

 One of these companies is Cambridge MA-based Werewolf Therapeutics that is advancing its pipeline of proinflammatory immune-modulating cytokines for cancer treatment. Two years after being founded in 2017, Werewolf came out of stealth mode with a $56 Million Series A in November 2019 followed by a $72 Million Series B in January 2021 led by RA Capital Management. Just three months later Werewolf went public with a $120 Million IPO valued at $440 Million to advance their products into IND filing. A later-stage company, California based ALX Oncology went public in July 2020 with a valuation of $668.8 Million, and just over a year later they have a current valuation of 2.64 Billion. ALX is advancing its CD47 checkpoint inhibitor, ALX148, through a number of clinical trials for a series of cancer indications.
One of these companies is Cambridge MA-based Werewolf Therapeutics that is advancing its pipeline of proinflammatory immune-modulating cytokines for cancer treatment. Two years after being founded in 2017, Werewolf came out of stealth mode with a $56 Million Series A in November 2019 followed by a $72 Million Series B in January 2021 led by RA Capital Management. Just three months later Werewolf went public with a $120 Million IPO valued at $440 Million to advance their products into IND filing. A later-stage company, California based ALX Oncology went public in July 2020 with a valuation of $668.8 Million, and just over a year later they have a current valuation of 2.64 Billion. ALX is advancing its CD47 checkpoint inhibitor, ALX148, through a number of clinical trials for a series of cancer indications.

2021 to date has seen 93 immuno-oncology deals totaling $7.9B, on par to compete with 111 deals in 2020. Additionally, nearly 38 companies have products in phase 2 trials or beyond, with hopes of being the next major treatment in the immuno-oncology field.